Model Chemistry¶
- All quantum chemistry calculations start with HF as a baseline
- Additional theories can be implemented:
- Møller-Plesset perturbation theory
- Configuration Interaction
- Coupled Cluster
- Additional theories can be implemented:
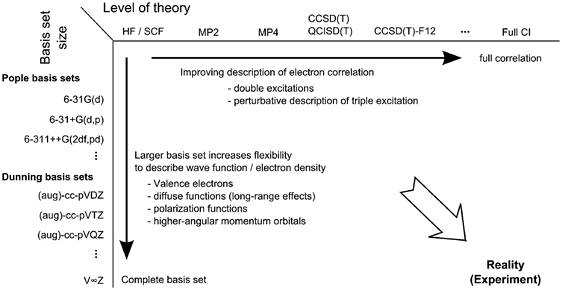
- Everything is a trade off between accuracy (large basis set, higher level of theory) and computational cost/time
- The higher the theory and basis set, the more realistic the results
- It’s worth noting that DFT
Electron Configuration * We define electron configuration in comp chem much like we do in physical chem * We use symmetry and number the order however, to describe the orbitals * E.g. for water:
\[
1(A_1 )^2 2(A_1 )^2 1(B_2 )^2 3(A_1 )^2 1(B_1 )^2 4(A_1 )^0 2(B_2 )^0
\]
- We use the terms HOMO and LUMO to describe these and can often describe the surrounding orbitals as HOMO-1 and LUMO+1
Koopman’s Theorem¶
- States that the ionisation energy of an atom or molecule is equal to the energy from the orbital of which the electron is ejected
\[
I_i=−\epsilon_t
\]
- In HF, the energies are more exactly calculated, however in DFT, we can only say that the energy is approximately equal to the ionisation energy (meta Koopman’s theory)
\[
I_i≈−\epsilon_t
\]
Coordinates¶
- The general rule of thumb is that the fewer coordinates there are, the less variables there are for the computer to have to optimise
- The connectivity is determined by the atom’s behaviour, so we don’t need to specify it explicitly
- We can depict geometry in two primary ways:
Cartesian Coordinates¶
- Use the cartesian system of X, Y and Z values for each atom
- E.g.
C x y z C x y z H x y z H x y z ...
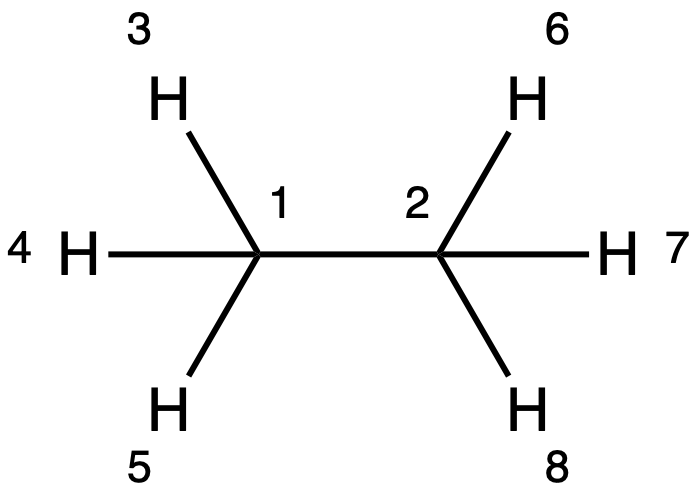
Z-Matrix¶
- Uses a system of internal coordinates of bond lengths, angles and dihedrals to define the geometry
- E.g.
C1 C2 R1 1 H3 R2 1 𝜃1 2 H4 R3 1 𝜃2 2 D1 6 H5 R4 1 𝜃3 2 D2 6 H6 R5 2 𝜃4 1 D3 3 H7 R6 2 𝜃5 1 D4 3 H8 R7 2 𝜃6 1 D5 3 R1= R2= ... 𝜃1= 𝜃2= ... D1= D2= ...